Polystyrene pieces are indispensable today, and palladium catalysts allowed their mass production. Source: image edited by @emiliomoron, original from pixabay.com.
The synthesis of many chemicals, especially pharmaceuticals and plastics, is based on the use of transition metals as catalysts at various points in their synthesis. And although these types of metals turn out to be efficient catalysts and some cases indispensable for many processes, unfortunately they are rare and toxic metals. In addition, ligands and other compounds are needed to control their reactivity and selectivity, so many times the metal catalyst is not recovered. This means that their use is unsustainable in the long term, so organic synthesis specialists need more sustainable alternatives.
That is why in a recent study published in Angewandte Chemie, researchers at the University of Kanazawa report a possible alternative. Conducting extensive research involving a large number of chemical synthesis reactions that are common in the pharmaceutical and petrochemical industry are paving the way for more sustainable chemistry without the use of transition metal catalysts in cross-coupling reactions.
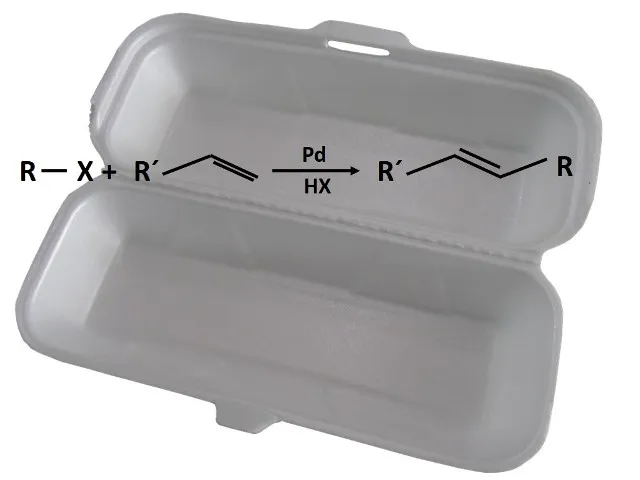
The principle of cross-coupling using transition metal catalysts is that two molecules are bonded to the metal through the formation of carbon-metal bonds, and in a second step, these carbon atoms are joined together and allow a new single carbon-carbon bond to be formed. These reactions marked a milestone in chemistry in the late 1970s, expanding the use of transition metals in synthesis reactions, especially in the synthesis of pharmaceuticals and agrochemicals.

Schematic form of a coupling reaction.
To offer an alternative, researchers at the University of Kanazawa used benzyl organoborates to perform tertiary alkyl electrophilic cross-linking. According to the study's author, the procedure does not require the use of rare metals and offers a direct route to quaternary carbon centers.
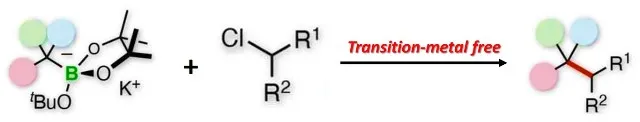
Cross-linking of alkyl or aryl electrophils in the tertiary sector.
The studied procedure consisted in the activation of a tertiary benzyl boronate by a base of potassium alkoxide to become a benzyl anion. This anion is then subjected to a cross-coupling reaction with a secondary alkyl chloride electrophilic. This type of reaction has a wide range, since the phenyl group of boronate can be replaced by several aromatic rings, and the electrophilic can be either an aromatic or a linear chain compound, which would allow the synthesis of various products by this route.
Further development of this type of procedure is essential. On the one hand, it is possible to reduce the dependence on transition metal-based catalysts, thus mitigating the environmental impact of their extraction, and on the other hand, the pandemic revealed the fragility of the rare metal supply chain, when a few months ago Japan reported that it would strengthen control over its rare metal reserves following the interruption in the supply of certain metals caused by the coronavirus pandemic.
Thanks for coming by to read friends, I hope you liked the information. See you next time.

